产品简介:
Cal-520™在探测细胞内钙离子方面提供了一个卓越的荧光分析工具。 Cal-520™ AM是新型的钙离子荧光染料相比现在还在应用的绿色钙离子荧光指示剂(例如 Fluo-3 AM 和 Fluo-4 AM)在信噪比和细胞内滞留方面有着显著的改进。G蛋白偶联受体或者.钙离子通道可以被 Cal-520™ AM 通过细胞膜标记钙离子而预先加载。 Cal 520™ AM 一旦进入细胞内,其亲脂基团就会被剪切酶水解成一个带阴性负离子的荧光染料被阻断在细胞内。它与钙离子结合其荧光就会显著的增强。当细胞内的细胞器受到刺激就会释放出钙离子, Cal-520™的荧光信号就会显著的增加。长波长、高灵敏性、大于100倍的荧光信号增强使得 Cal-520™ AM成为一个理想的细胞内钙离子测量指示器。高的信噪比和极好的细胞内滞留使得Cal-520™在测评 GPCR 和钙离子通道目标及他们的受体激动剂和拮抗剂的筛选方面相比其他钙离子探针显现更加卓越的优越性。相比其他 Cal-520™指示器 Cal-520FF™具有最小的钙离子亲和力他用以标记 Kd ~ 10 uM单位的钙离子。
| 规格 | 1 mg | 产品形式 | 粉末 |
| Ex (nm) | 492 | Em (nm) | 514 |
| 分子量 | 1138.92 | 溶剂 | DMSO |
储存条件:
-20℃
使用方法推荐(仅供参考)
- 1. Load Cells with Cal-520®,AM:
- AM esters are non-polar esters that can readily cross live cell membranes, and rapidly hydrolyzed by cellular esterases inside live cells. AM esters are widely used for loading a variety of polar fluorescent probes into live cells noninvasively. However, cautions must be exercised when AM esters are used since they are susceptible to hydrolysis, particularly in solution. They should be reconstituted just before use in high-quality, anhydrous dimethylsulfoxide (DMSO). DMSO stock solutions may be stored desiccated at –20 °C and protected from light. Under these conditions, AM esters should be stable for several months. Following is our recommended protocol for loading Cal-520® AM,Cal-590™ AM or Cal-630™ AM esters into live cells. This protocol only provides a guideline, and should be modified according to your specific needs.
-
- Prepare a 2 to 5 mM stock solution of Cal-520® AM in high-quality, anhydrous DMSO.
- On the day of the experiment, either dissolve Cal-520® AM, in DMSO or thaw an aliquot of the indicator stock solution to room temperature. Prepare a dye working solution of 10 to 20 µM in Hanks and Hepes buffer (HHBS) or the buffer of your choice with 0.04%Pluronic® F-127. The exact concentration of the indicator required for cell loading must be determined empirically.
Note:The nonionic detergent Pluronic® F-127 is sometimes used to increase the aqueous solubility of Cal-520® AM,. - If your cells (such as CHO cells) contain organic anion-transports, then probenecid (1-2 mM) may be added to the dye working solution (final in well concentration will be 0.5-1 mM) to reduce leakage of the de-esterified indicators.
- Add equal volume of the dye working solution (from Step b or c) into your cell plate.
- Incubate the dye-loading plate in a cell incubator for 60 to 90 minutes, and then incubate the plate at room temperature for another 30 minutes.
Note: Incubating the dye longer than 2 hours gives better signal intensity for some cell lines.
- Replace the dye working solution with HHBS or if applicable, a buffer of your choice that contains an anion transporter inhibitor, such as 1 mM probenecid, to remove excess probes.
- Run the experiments at Ex/Em = 490/525 nm (for Cal-520® AM), 540/590 nm
-
- 2. Measure Intracellular Calcium Responses:

Figure 1.Response of endogenous P2Y receptor to ATP in CHO-M1 cells withoutprobenecid.CHO-M1 cells were seeded overnight at 40,000 cells per 100 µL per well in a 96-well black wall/clear bottom costar plate. 100 µl of 4 µM Fluo-3 AM, Fluo-4 AM or Cal 520® AM in HHBS were added into the wells, and the cells were incubated at 37 °C for 2 hour. The dye loading medium were replaced with 100 µl HHBS, 50 µl of 300 µM ATP were added, and then imaged with a fluorescence microscope (Olympus IX71) using FITC channel.
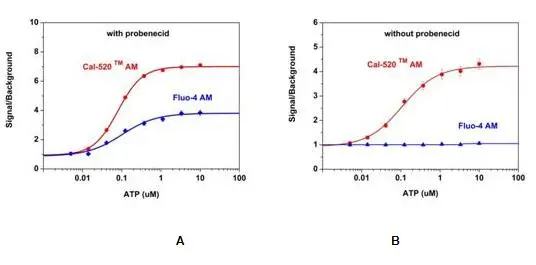
Figure 2.ATP-stimulated calcium response of endogenous P2Y receptor in CHO-K1 cells measured with Cal-520® or Fluo-4 AM.CHO-K1cells were seeded overnight in 50,000 cells per 100 µL per well in a 96-well black wall/clear bottom costar plate. 100 µL of 5 µM Fluo-4 AM or the Cal-520® AM with (A) or without (B) 2.5 mM probenecid was added into the cells, and the cells were incubated at 37oC for 2 hours. ATP (50µL/well) was added by FlexStation (Molecular Devices) to achieve the final indicated concentrations.
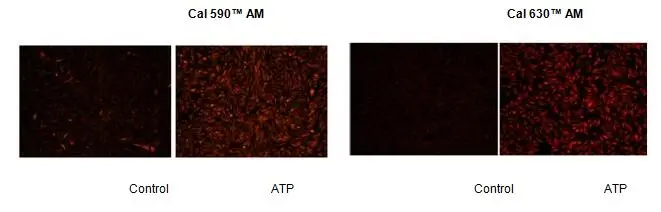
Figure 3.Response of endogenous P2Y receptor to ATP in CHO-K cells.CHO-K cells were seeded overnight at 40,000 cells per 100 µL per well in a 96-well black wall/clear bottom costar plate. 100 µl of 4 µM Cal 590™ AM or Cal 630™ AM in HHBS with 1 mM probenecid were added into the wells, and the cells were incubated at 37 °C for 2 hour. The dye loading mediums were replaced with 100 µl HHBS and 1 mM probenecid , then imaged with a fluorescence microscope (Olympus IX71) using TRITC channel before and after adding 50 µl of 300 µM ATP.



